|
2/16/2024 0 Comments Cobalt oxidation number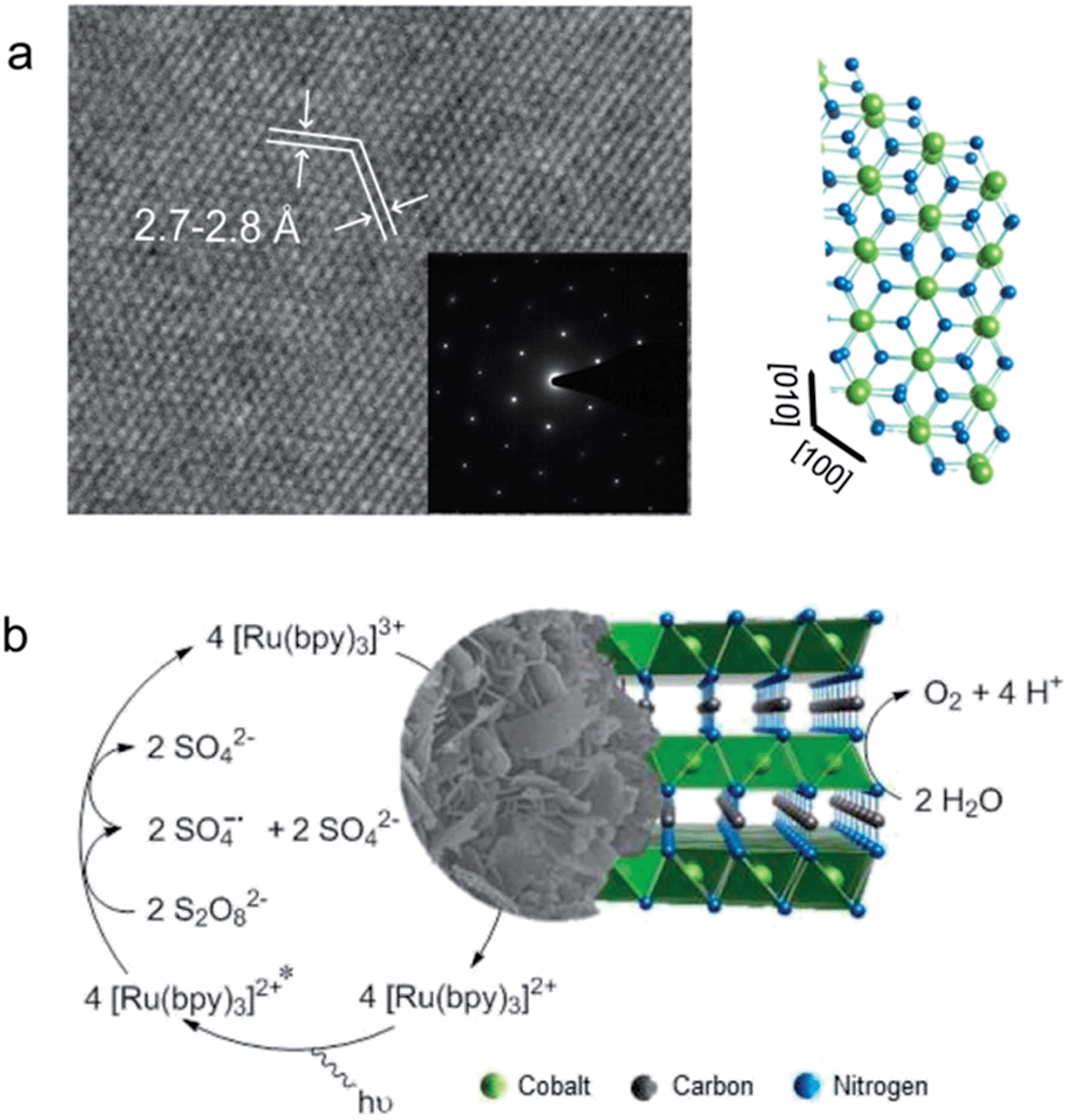
These active sites in a reduced coordinate environment possess unique electronic and structural characteristics, which are found beneficial for the charge-transfer and adsorption of intermediates during the electrochemical reaction 16, 17, 18. Active transition-metal with lower coordination numbers (in trigonal bipyramidal geometry) has also been reported with a coordination number switching between 4 and 5 during the OER 16, 17. The OER active site in octahedral geometry (or square pyramidal geometry for an undercoordinated surface site) has been dominant and is generally regarded as a promising local structure with a coordination number switching between 5 and 6 during the OER 13, 14, 15. However, the design of advanced catalysts with high activity and sustainability is still challenging owing to limited choices 11, 12. In the last years, various transition-metal-based complex compounds have been intensively explored for catalyzing the OER efficiently 1, 2, 3, 4, 5, 6, 7, 8, 9, 10. The complex four-electron transfer process makes OER occurs at a large overpotential and thus generally consumes considerable energy. In these systems, the oxygen evolution reaction (OER) is an essential step. 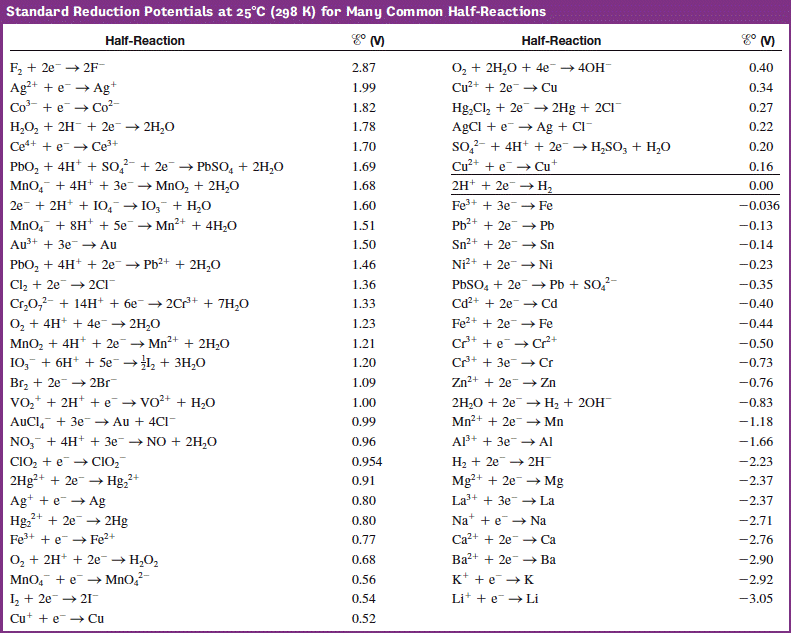
Meanwhile, several eco-friendly energy conversion systems, such as water electrolyzers and metal-air batteries, have been explored to store energy from renewable resources. Similar content being viewed by othersĪ sustainable society relies upon renewable energy resources instead of fossil fuel combustion to generate electricity, reducing emissions of pollutants. We expect that the reported active structural motif of dual corner-shared cobalt tetrahedra in this study could enable further development of compounds for catalyzing the OER. The density functional theory calculations demonstrate that the OER is efficiently catalyzed by a binuclear active site of dual corner-shared cobalt tetrahedra, which have a coordination number switching between 3 and 4 during the reaction. The bulk of YBaCo 4O 7 composes of corner-sharing only CoO 4 tetrahedra, which can flexibly alter their positions to accommodate the insertion of interstitial oxygen ions and mediate the stress during the electrochemical oxidation. We reveal that the surface of YBaCo 4O 7 possesses strong resilience towards structural amorphization during OER, which originates from its distinctive structural evolution toward electrochemical oxidation. Here, we report that the oxygen evolution reaction (OER) can be efficiently catalyzed by cobalt tetrahedra, which are stabilized over the surface of a Swedenborgite-type YBCo 4O 7 material. Developing efficient catalysts is of paramount importance to oxygen evolution, a sluggish anodic reaction that provides essential electrons and protons for various electrochemical processes, such as hydrogen generation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
